A battery works when electricity is produced by something called an oxidation/reduction (redox) reaction.
In this project, you and your child (adult supervision is required) use two different metals and some salty liquid to create a redox reaction. One metal will oxidize and lose electrons. The other will reduce and gain electrons in the process. These reactions can result produce toxic chemicals, so use these precautions:
- Wash hands before and after experimenting with batteries and electrolytic cells.
- Experiment only in a well-ventilated space.
- Wear latex or nitrile gloves.
- Do not reuse containers you use in this project for food or other non-science purposes.
What Does That Mean?
A redox reaction is a chemical reaction involving the transfer of electrons. It's what's happening when metal rusts, fires start, photosynthesis happens in plants, and we breathe by inhaling oxygen and exhaling carbon dioxide.
Materials & Tools
Water
Salt
Nonmetallic jar or bowl (glass or plastic)
Different types of metal objects: paper clips, coins, nails and screws, etc.

Alligator Clips
Voltmeter
Directions
Step 1: Make Salt Water
Pour 2 cups of water into your jar or bowl. Pour 1 tbsp. of salt into the water and mix until dissolved.
Step 2: Set Up Your Leads
Connect one alligator clip to one metal object and connect a second clip to a second metal object.

Dip the objects into the salt water. Don't let the alligator clips touch the salt water.
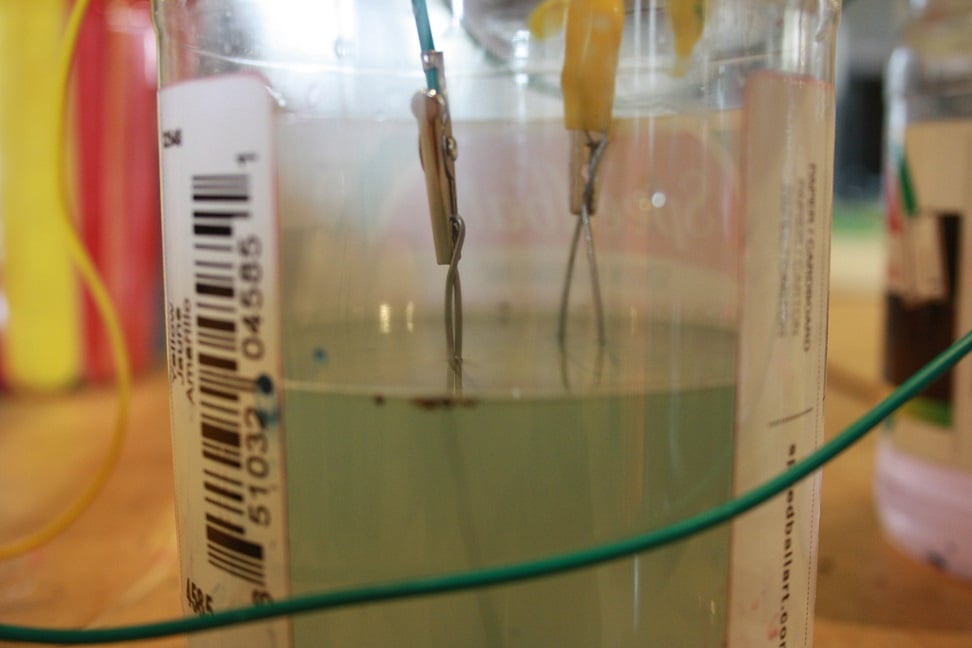
Step 3: Testing
Attach the clips to your volt meter and see how much voltage the battery is producing.

Step 3: Iteration
Try different combinations of metals and liquids to see what results you get.